It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
DP
edit on am25America/ChicagoSaturday2022-02-05T05:38:01-06:0005America/Chicago02000000 by angelchemuel because: (no reason given)
a reply to: angelchemuel
No ones talking about forcing the vaccine on anyone in Africa or Asia.
Just to let those who want to have hit, get it.
We haven't been doing a very good job so far, and there should be overproduction by now.
No ones talking about forcing the vaccine on anyone in Africa or Asia.
Just to let those who want to have hit, get it.
We haven't been doing a very good job so far, and there should be overproduction by now.
a reply to: MonkeyBalls2
perhaps they don't need it due to good immune systems, Ivermectin regularly used, plenty of UVB to produce Vit D and other benefits.
Plus a dam site worst diseases and bugs to worry about.
perhaps they don't need it due to good immune systems, Ivermectin regularly used, plenty of UVB to produce Vit D and other benefits.
Plus a dam site worst diseases and bugs to worry about.
originally posted by: MonkeyBalls2
a reply to: angelchemuel
No ones talking about forcing the vaccine on anyone in Africa or Asia.
Just to let those who want to have hit, get it.
We haven't been doing a very good job so far, and there should be overproduction by now.
Sorry to tell ya, but Hong Kong now is telling even foreigners now must be vaccinated with the sinovax ( whatever they call it ) Suppossely it not the same crap as Moderna.
So who remembers the covid Human Challenge trials in the UK?
Apparently the infected 36 people.
Fully vetted no vaccine, no evidence of previous infection and no comorbidities.
18 infected sero-negative
16 uninfected sero-negative
2 Uninfected sero-positive
So 16 dealt with virus without any effect and didn't generate anti-bodies the innate immune system just shut it down
2 dealt with it in 2 days generating antibodies.
Proof showing the remdesivir shows no benefit and was stopped. Yet is still on the NHS prevention offers.
Apparently the infected 36 people.
Fully vetted no vaccine, no evidence of previous infection and no comorbidities.
18 infected sero-negative
16 uninfected sero-negative
2 Uninfected sero-positive
So 16 dealt with virus without any effect and didn't generate anti-bodies the innate immune system just shut it down
2 dealt with it in 2 days generating antibodies.
Proof showing the remdesivir shows no benefit and was stopped. Yet is still on the NHS prevention offers.
Been a while since a piccy update
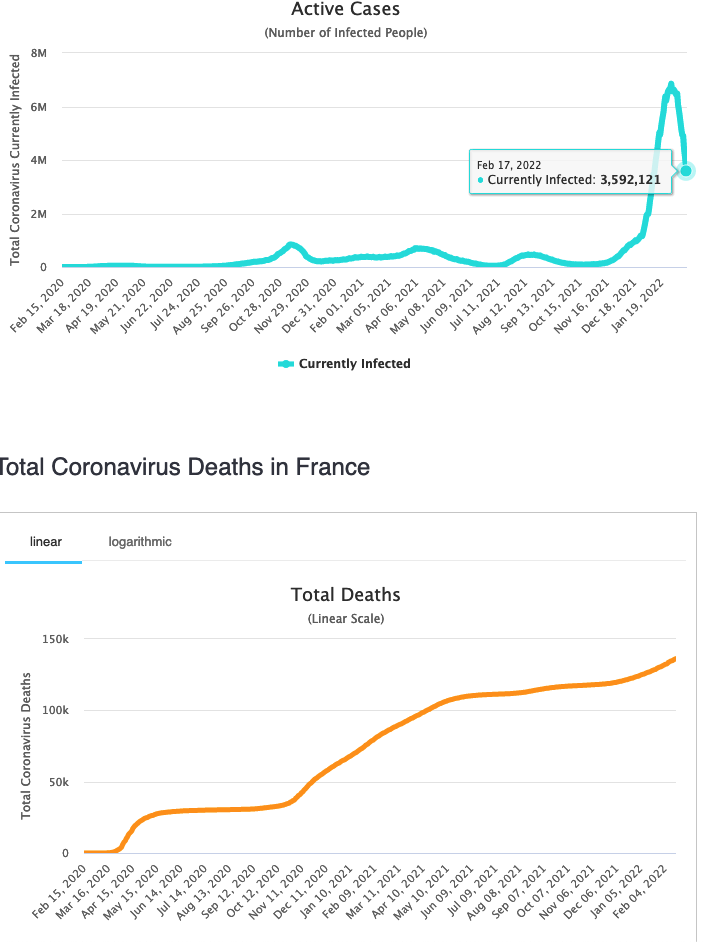
France Update : Infections are falling, but Deaths are still climbing.
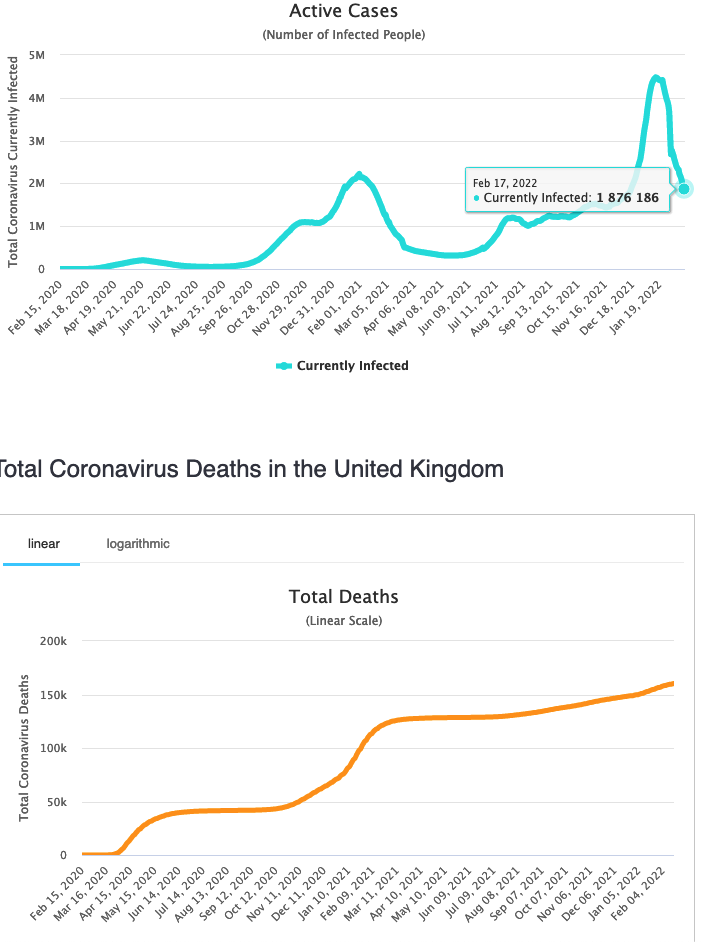
UK is doing about the same.
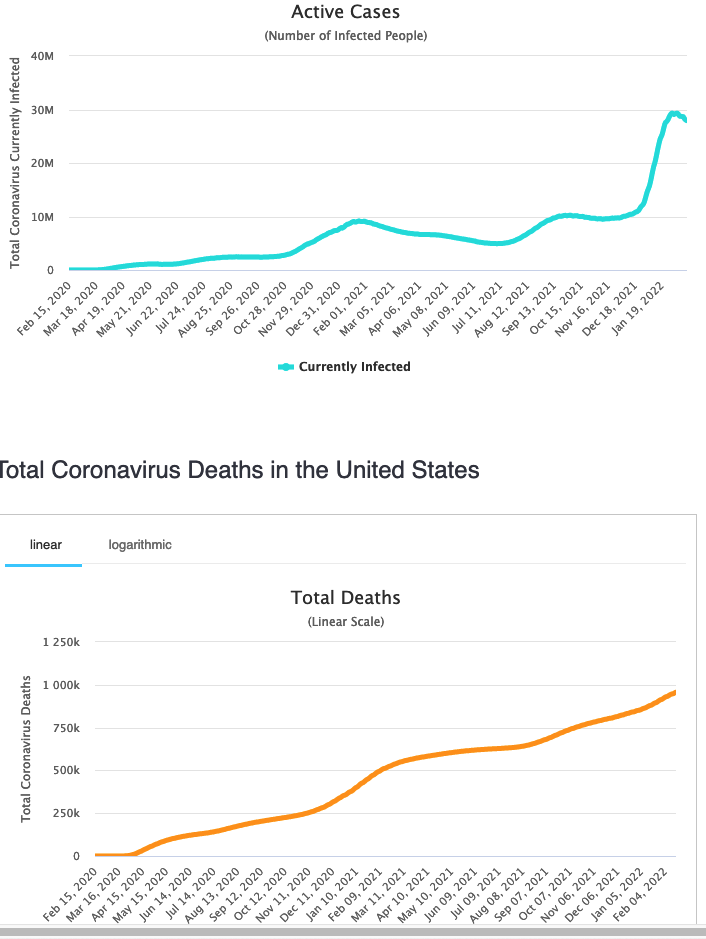
USA infections are turning the peak, but Deaths are climbing still.
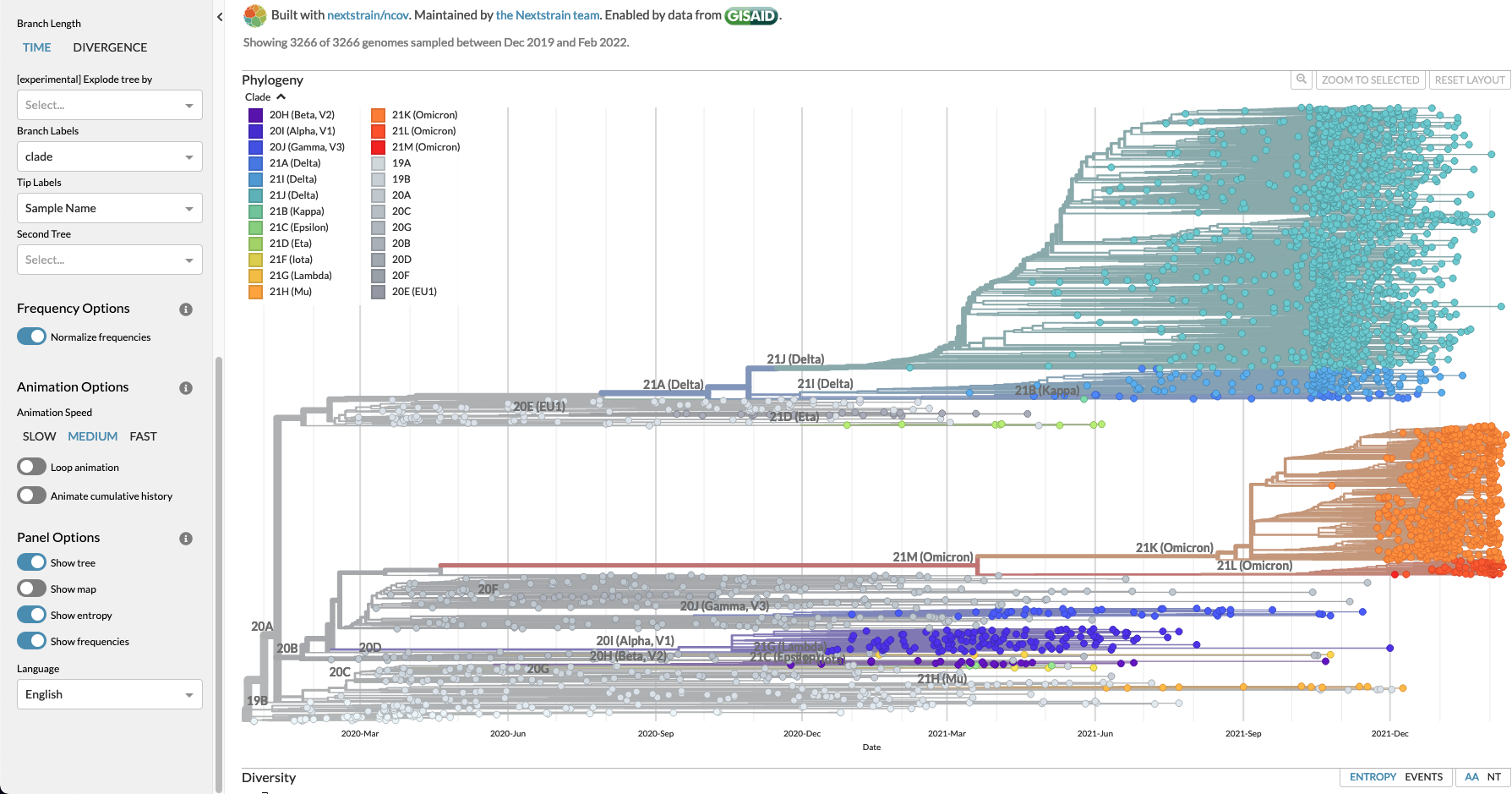
and finally an update from NextStrain.org.
As an added bonus, due to the current political climate in the world, I've added links in my sig below to a couple of films from the early 80's that are worth the watch.
France Update : Infections are falling, but Deaths are still climbing.

UK is doing about the same.

USA infections are turning the peak, but Deaths are climbing still.

and finally an update from NextStrain.org.

As an added bonus, due to the current political climate in the world, I've added links in my sig below to a couple of films from the early 80's that are worth the watch.
a reply to: MonkeyBalls2
Thanks for the updates! Probably makes sense for the deaths to stay on the inclination a little while after infection peak, but we should see them fall just as quickly as infection very soon (I think!). If not then vaccine death has to be considered as playing a greater role than we’re told. Hoping not, but if that were the case, then expect a new variant announcement as cover story for the continuing death rate, bringing infection rates back up artificially. I think a clue to that will be if deaths don’t dip at all between variants (not proof, but very convenient timing).
As for that other little sideshow apocalypse you mentioned, someone should strap Putin and Biden (and their generals) to Clockwork Orange chairs, eyelids held open with those links in your signature playing on loop for a few days.
I’m old enough to recall the early 80s when nuclear conflict hung in the air like it does now. But then there seemed to be a straight forward approach to avoidance. Now, thanks to the Information age and it’s fake news there’s magnitudes more bluff and spin playing out that I fear even those in charge don’t really understand. Plus there’s a strange lack of public engagement with the existential threat, thanks no doubt to being distracted by the virus. The world could end and no one would notice, busy watching Netflix and hashtagging until of course that shockwave hit.
There’s so many narratives around these days I don’t think people can tell apart the ones that matter anymore.
Thanks for the updates! Probably makes sense for the deaths to stay on the inclination a little while after infection peak, but we should see them fall just as quickly as infection very soon (I think!). If not then vaccine death has to be considered as playing a greater role than we’re told. Hoping not, but if that were the case, then expect a new variant announcement as cover story for the continuing death rate, bringing infection rates back up artificially. I think a clue to that will be if deaths don’t dip at all between variants (not proof, but very convenient timing).
As for that other little sideshow apocalypse you mentioned, someone should strap Putin and Biden (and their generals) to Clockwork Orange chairs, eyelids held open with those links in your signature playing on loop for a few days.
I’m old enough to recall the early 80s when nuclear conflict hung in the air like it does now. But then there seemed to be a straight forward approach to avoidance. Now, thanks to the Information age and it’s fake news there’s magnitudes more bluff and spin playing out that I fear even those in charge don’t really understand. Plus there’s a strange lack of public engagement with the existential threat, thanks no doubt to being distracted by the virus. The world could end and no one would notice, busy watching Netflix and hashtagging until of course that shockwave hit.
There’s so many narratives around these days I don’t think people can tell apart the ones that matter anymore.
a reply to: McGinty
There's a good chance the vaccine is preventing Deaths, but it's not something you can really prove without a doubt
We could be lucky, and this will be the last of it, but being the pessimist, and it being 'all over the world' still, yet again, the probabilities of another variant coming to the fore, is above 100%.
We might get a Spring-Summer reprieve i think, but that depends on people not bringing it over from area's where it will be winter.
As per usual, its a waiting game...
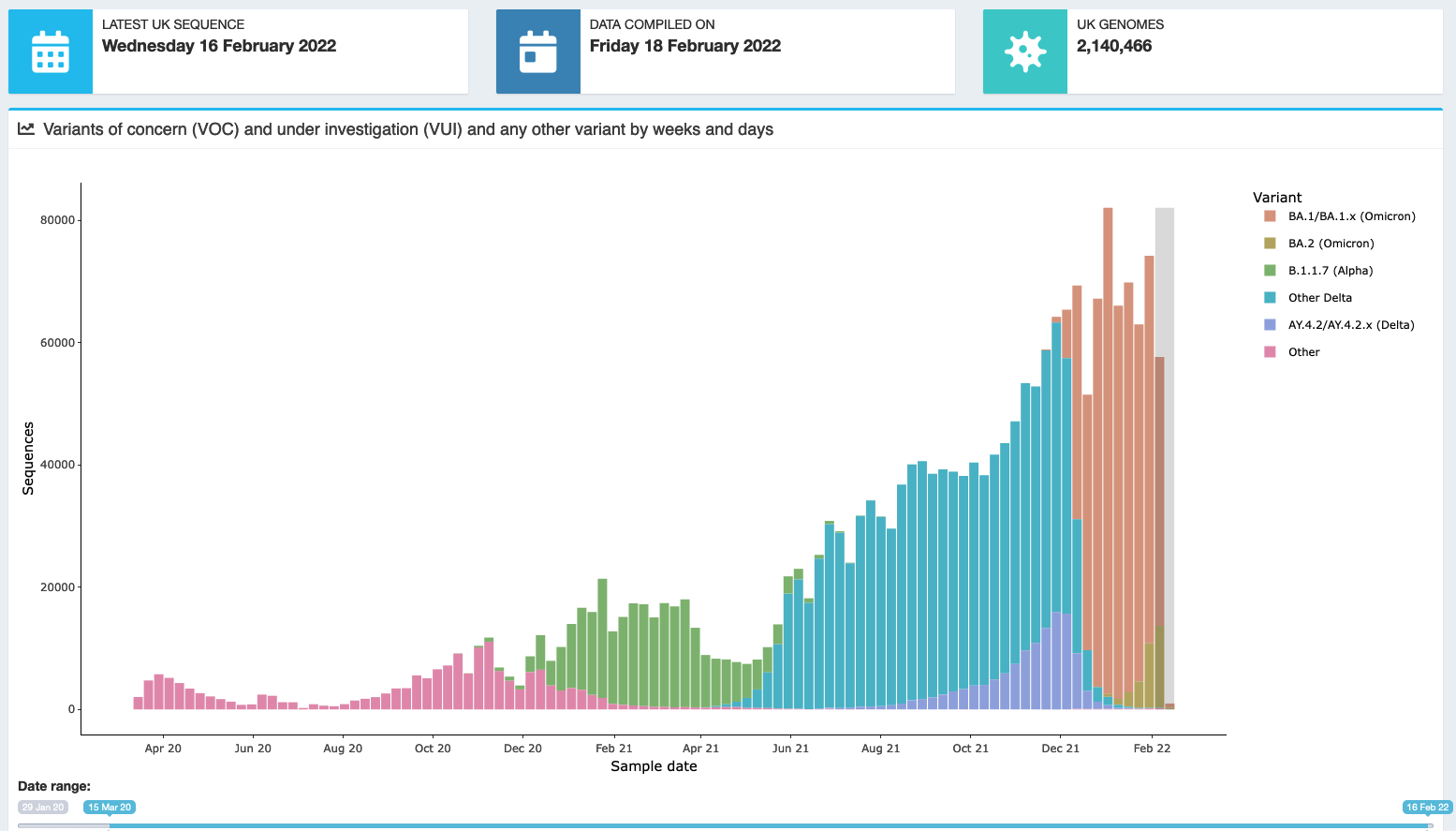
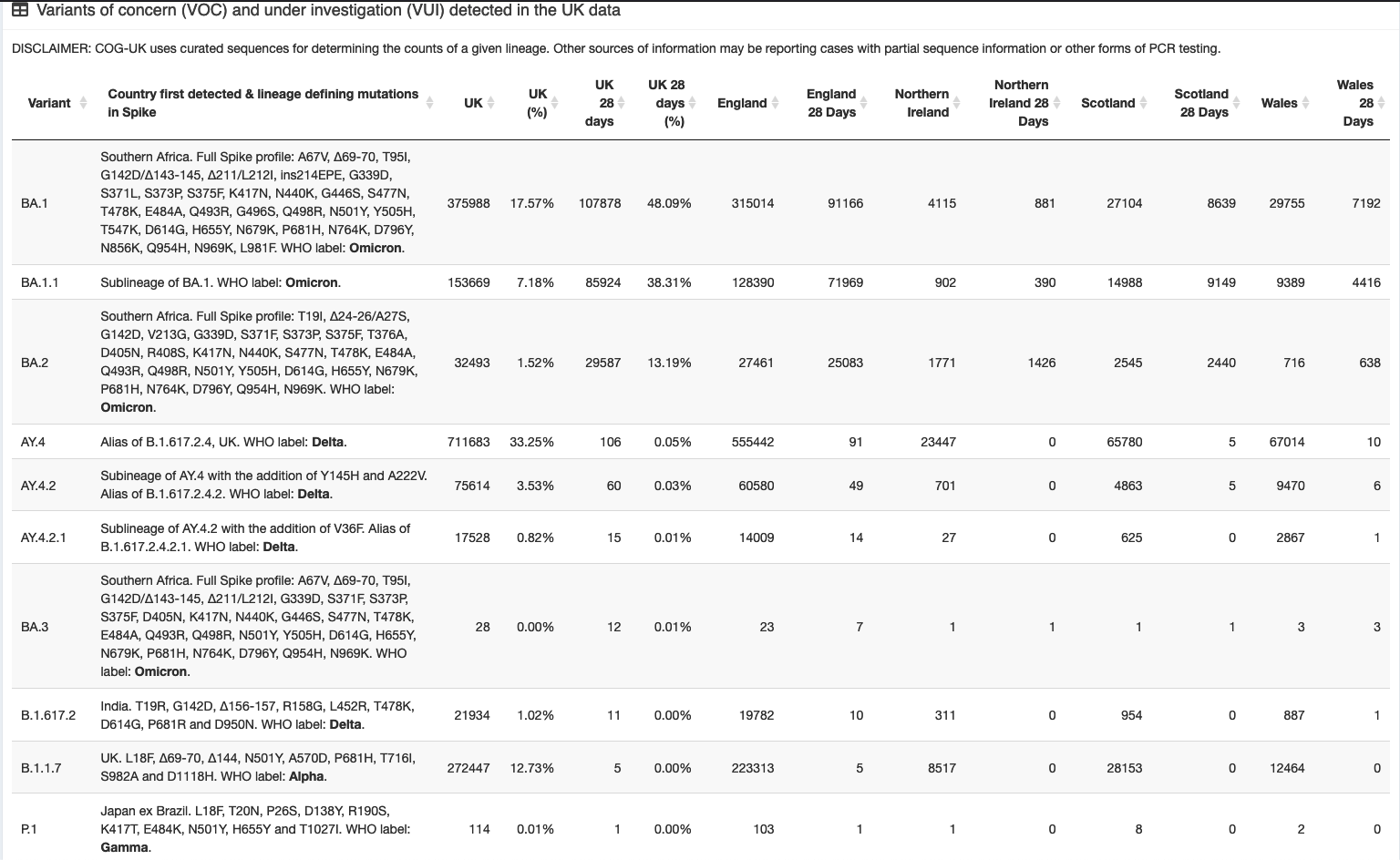
Here's a couple of caps from the COG-UK Site, you can see that BA.1.1 and BA.2 have been starting to get noticed over the last month or two, getting into their stride.
It was mentioned somewhere (need to find a link) that BA.2 was possibly as infectious as original Omicron, but had similar lung infections as Delta, instead of throat like BA.1 Omicron (Is this the Deltacron ?)
It has yet to be seen if it is as dangerous though (That 'time' thing again...) :
I removed the "Threads" link, as it turned out to be a fake (should have checked before posting, but it was late).
Can't seem to find a proper Youtube link to it...
Lets just hope that cooler heads prevail.
'The Day After' apparently affected Reagan quite a lot at the time...so maybe you're right about the Clockwork Orange chair
(I'd leave Boris in the chair, so his Ex's and many, many children, could kick him in the gonads for as long as they like, and then let the general populace have a go. I'd pay a Fiver to get a 'kick' out of it.
"Roll up! Roll up!" LOL take him on a tour of the Kingdom )
edit : As a mea-culpa for 'Threads', here's a link to a couple more vids from back in the day (Watched them last night) :
Panorama - If The Bomb Drops (1980 Nuclear War episode, precursor to 'Threads')
and
On the 8th Day - Nuclear Winter Documentary (1984)
There's a good chance the vaccine is preventing Deaths, but it's not something you can really prove without a doubt
We could be lucky, and this will be the last of it, but being the pessimist, and it being 'all over the world' still, yet again, the probabilities of another variant coming to the fore, is above 100%.
We might get a Spring-Summer reprieve i think, but that depends on people not bringing it over from area's where it will be winter.
As per usual, its a waiting game...
Here's a couple of caps from the COG-UK Site, you can see that BA.1.1 and BA.2 have been starting to get noticed over the last month or two, getting into their stride.
It was mentioned somewhere (need to find a link) that BA.2 was possibly as infectious as original Omicron, but had similar lung infections as Delta, instead of throat like BA.1 Omicron (Is this the Deltacron ?)
It has yet to be seen if it is as dangerous though (That 'time' thing again...) :


I removed the "Threads" link, as it turned out to be a fake (should have checked before posting, but it was late).
Can't seem to find a proper Youtube link to it...
Lets just hope that cooler heads prevail.
'The Day After' apparently affected Reagan quite a lot at the time...so maybe you're right about the Clockwork Orange chair
(I'd leave Boris in the chair, so his Ex's and many, many children, could kick him in the gonads for as long as they like, and then let the general populace have a go. I'd pay a Fiver to get a 'kick' out of it.
"Roll up! Roll up!" LOL take him on a tour of the Kingdom )
edit : As a mea-culpa for 'Threads', here's a link to a couple more vids from back in the day (Watched them last night) :
Panorama - If The Bomb Drops (1980 Nuclear War episode, precursor to 'Threads')
and
On the 8th Day - Nuclear Winter Documentary (1984)
edit on 19-2-2022 by MonkeyBalls2 because: added edit
a reply to: McGinty
The only way we will get Zero covid deaths is if people stopped dying of any cause in the UK
"Weekly deaths with COVID-19 on the death certificate by date registered" and " whose death certificate mentioned COVID-19 as one of the causes"
Number people who died within 28 days of being identified as a COVID-19 case by a positive test.
Without clarification they will include all Omicron cases as a "covid" death regardless of actual death.
The only way we will get Zero covid deaths is if people stopped dying of any cause in the UK
"Weekly deaths with COVID-19 on the death certificate by date registered" and " whose death certificate mentioned COVID-19 as one of the causes"
Number people who died within 28 days of being identified as a COVID-19 case by a positive test.
Without clarification they will include all Omicron cases as a "covid" death regardless of actual death.
a reply to: puzzled2
It’s amazing how history can be littered with families and corporations profiteering from war and suffering yet so many people seem to think “No, not this time - this time it’s different”.
Perhaps they are shills, perhaps not, but even here on a conspiracy website there are people that seem to think our leaders would never dream of prioritising the needs of the few over the many. If ATS has such vociferous support for TPTB, then just imagine how deeply seeded that denial or naivete must be in the Sheeple?
Covid is perhaps the biggest business opportunity in the history of the human race. Does anyone really believe that there are not queues of pharma reps at the doors of every PM around the globe petitioning them with every means at their disposal to maximise vaccine sales and potential vaccine after-care?
Every generation appears to have no shortage of suckers.
Note: The question of covid’s origin and pathology is besides the above point. Whether manufactured, escaped, released or an accident of nature now that it’s here covid represents a huge, airborne dollar sign.
It’s amazing how history can be littered with families and corporations profiteering from war and suffering yet so many people seem to think “No, not this time - this time it’s different”.
Perhaps they are shills, perhaps not, but even here on a conspiracy website there are people that seem to think our leaders would never dream of prioritising the needs of the few over the many. If ATS has such vociferous support for TPTB, then just imagine how deeply seeded that denial or naivete must be in the Sheeple?
Covid is perhaps the biggest business opportunity in the history of the human race. Does anyone really believe that there are not queues of pharma reps at the doors of every PM around the globe petitioning them with every means at their disposal to maximise vaccine sales and potential vaccine after-care?
Every generation appears to have no shortage of suckers.
Note: The question of covid’s origin and pathology is besides the above point. Whether manufactured, escaped, released or an accident of nature now that it’s here covid represents a huge, airborne dollar sign.
edit on 22-2-2022 by McGinty because: (no reason given)
Veterinarians have been doing this for decades, that's why it's called 'herd immunity'. I mentioned this when the vaccines started rolling out last
year.
"no documented studies yet".... yeah.... none that us the public will be privy too!
Look who one of the main funders of the research is...
Report
Rainbows
Jane
"While there are no documented studies yet involving self-spreading vaccines for use in humans, even before the coronavirus pandemic struck, it was something UK government officials were exploring as a viable option for tackling seasonal flu.
In a 2019 paper prepared for the Department of Health and Social Care, officials mapped out a hypothetical case to show how releasing a self-spreading flu vaccine into the UK population could be used to reduce illness and save lives."
"no documented studies yet".... yeah.... none that us the public will be privy too!
Look who one of the main funders of the research is...
The research is being subsidised by high-profile funding organisations, including the U.S. National Institutes of Health (the country’s main medical research agency).
Report
Rainbows
Jane
Good news we have a Plant based Vaccine approved in Canada that is only 20% less effective as the natural immunity found in most healthy or
Asymptomatic humans.
source
source
Medicago and GlaxoSmithKline announced that Health Canada has granted approval for Covifenz, COVID-19 vaccine, (plant-based virus-like particles [VLP], recombinant, adjuvanted). This vaccine is indicated for active immunization to prevent COVID‑19 caused by severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2) in individuals 18 to 64 years of age. The vaccination regimen calls for two doses given intramuscularly 21 days apart.
The Canadian approval of Covifenz was based on positive phase III data which showed that Covifenz demonstrated 71% efficacy against all variants of the virus. Against the Gamma variant, the vaccine was 88.6% effective, and 75.3% effective against the Delta variant. No cases of the Alpha, Lambda and Mu variants were observed in the vaccinated group while 12 cases were observed in the placebo group, the companies said. The Omicron variant was not circulating during the time the study was being conducted, both GSK and Medicago said.
Medicago and GSK submitted data to Health Canada on a rolling submission beginning in April 2021.
edit on 27-2-2022 by puzzled2 because: Changed source as it didn't link properly
a reply to: musicismagic
Well that is a country that I will not be visiting any time soon if not ever, this people just love pushing the darn mRNA gene therapy anyway they can.
Well that is a country that I will not be visiting any time soon if not ever, this people just love pushing the darn mRNA gene therapy anyway they can.
a reply to: marg6043
Still not understanding mRNA technology?
As you posted to me in another thread, ridiculing me for not having a medical qualification when I dared to express an opinion, may I respectfully ask if you have any medical qualification to support your opinion?
Still not understanding mRNA technology?
As you posted to me in another thread, ridiculing me for not having a medical qualification when I dared to express an opinion, may I respectfully ask if you have any medical qualification to support your opinion?
new topics
-
Ex-Border Patrol Chief Testifies that Biden/Harris Ordered Media Cover Up!
US Political Madness: 22 minutes ago -
A sheriff has been arrested after shooting at a judge at county courthouse with a fatality
Other Current Events: 40 minutes ago -
A small potential issue with Kamala.
US Political Madness: 44 minutes ago -
Oklahoma announced that they have removed 450,000 ineligible names from the voter rolls
2024 Elections: 56 minutes ago -
Did you vote are you gonna vote....
Jokes, Puns, & Pranks: 1 hours ago -
Israel Launches 2nd Wave Of EXPLODING TECH, Hezbollah Radios Begin Exploding
World War Three: 5 hours ago -
It is EASY for Illegal Aliens to Vote in Key Swing States and Not Have Their Ballots Rejected.
2024 Elections: 5 hours ago -
Nigel Farage : I am giving up control of the party to its members.
Regional Politics: 7 hours ago -
I want Love!
Rant: 7 hours ago -
Aerospace’s Secret Search For Antigravity
Science & Technology: 8 hours ago
top topics
-
20 Supporters Seated Behind Trump Onstage Rushed to Hospital
2024 Elections: 11 hours ago, 25 flags -
FBI says Iran sent hacked Trump info to Biden campaign
US Political Madness: 12 hours ago, 13 flags -
Israel Launches 2nd Wave Of EXPLODING TECH, Hezbollah Radios Begin Exploding
World War Three: 5 hours ago, 10 flags -
It is EASY for Illegal Aliens to Vote in Key Swing States and Not Have Their Ballots Rejected.
2024 Elections: 5 hours ago, 10 flags -
There would be no political madness, if We the People know the Truth
US Political Madness: 14 hours ago, 9 flags -
Limits on Technology
Science & Technology: 9 hours ago, 9 flags -
Gavin Newsom passes law against deepfake political ads
Politicians & People: 17 hours ago, 8 flags -
I want Love!
Rant: 7 hours ago, 6 flags -
Aerospace’s Secret Search For Antigravity
Science & Technology: 8 hours ago, 4 flags -
A sheriff has been arrested after shooting at a judge at county courthouse with a fatality
Other Current Events: 40 minutes ago, 3 flags
active topics
-
A sheriff has been arrested after shooting at a judge at county courthouse with a fatality
Other Current Events • 2 • : putnam6 -
UK Channel 4 programme The Trump Heist
Mainstream News • 82 • : Justoneman -
Israel Launches 2nd Wave Of EXPLODING TECH, Hezbollah Radios Begin Exploding
World War Three • 19 • : TzarChasm -
Ex-Border Patrol Chief Testifies that Biden/Harris Ordered Media Cover Up!
US Political Madness • 1 • : TheLieWeLive -
A small potential issue with Kamala.
US Political Madness • 4 • : xuenchen -
Gavin Newsom passes law against deepfake political ads
Politicians & People • 25 • : BingoMcGoof -
Limits on Technology
Science & Technology • 24 • : Terpene -
Why Donald J Trump is a threat to Democracy and Normal Feedoms
US Political Madness • 73 • : RazorV66 -
Oklahoma announced that they have removed 450,000 ineligible names from the voter rolls
2024 Elections • 3 • : xuenchen -
-@TH3WH17ERABB17- -Q- ---TIME TO SHOW THE WORLD--- -Part- --44--
Dissecting Disinformation • 2665 • : RelSciHistItSufi
