It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
a reply to: puzzled2
The way this is panning out is that if you get a few jabs and survive the initial spike in deaths, the initial vaccine reactions settle down, then you have a falling T cell problem to deal with, which in the end will cause inflammation and various personal troubles, but wait for theirs more they have a rebranded AIDs drug which will take care of this. This seems to be following modern medical practice, of going to the doc for a problem and then having to take a handful of petrochemical-derived drugs to counter the side effect of the original one. Which just mask and manage the illness. Tyranny comes in many guises. It's drug dealers' ply to get you hooked one way or another.
The way this is panning out is that if you get a few jabs and survive the initial spike in deaths, the initial vaccine reactions settle down, then you have a falling T cell problem to deal with, which in the end will cause inflammation and various personal troubles, but wait for theirs more they have a rebranded AIDs drug which will take care of this. This seems to be following modern medical practice, of going to the doc for a problem and then having to take a handful of petrochemical-derived drugs to counter the side effect of the original one. Which just mask and manage the illness. Tyranny comes in many guises. It's drug dealers' ply to get you hooked one way or another.
Phaahla and Professors Tulio de Oliveira and Richard Lascelles confirmed the detection of variant B.1.1.529 during an "urgent" briefing on Thursday
afternoon.
So will this get it's own name or just be Delta ++++
Can not be a South African variant be cause as usual no idea of the origin or why is suddenly mutated 52 changes on the spike.
Perhaps if we don't hurry up and follow orders they will be forced to release a new variant that causes in more cases.
Forgetting the mRNA injections will the other vaccines that use the fully viral particle be more effective? Will those that have had a proper case have the better immune system?
Lots of questions.
So will this get it's own name or just be Delta ++++
Can not be a South African variant be cause as usual no idea of the origin or why is suddenly mutated 52 changes on the spike.
Perhaps if we don't hurry up and follow orders they will be forced to release a new variant that causes in more cases.
Forgetting the mRNA injections will the other vaccines that use the fully viral particle be more effective? Will those that have had a proper case have the better immune system?
Lots of questions.
a reply to: puzzled2
I saw last night it was supposed to be called 'Nu', but this morning it seems to be called 'Omicron'.
BBC Website
The Guardian Article
edit : Could explain why France's numbers are going up so fast.
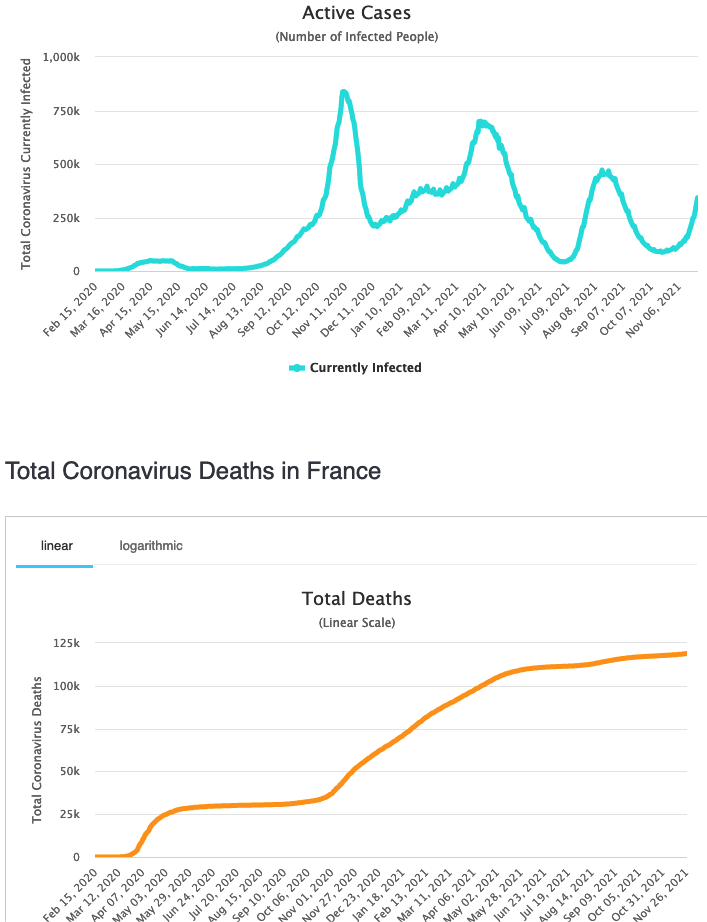
Quite a lot of people of African origin in France, and they don't tend to like being vaccinated.
I'm not generalizing, one of my colleagues if is from Togo, and he prefers 'Grandma's Remedy'...
Another colleague is also from Togo, and he's double vaxxed.
I saw last night it was supposed to be called 'Nu', but this morning it seems to be called 'Omicron'.
BBC Website
The Guardian Article
edit : Could explain why France's numbers are going up so fast.

Quite a lot of people of African origin in France, and they don't tend to like being vaccinated.
I'm not generalizing, one of my colleagues if is from Togo, and he prefers 'Grandma's Remedy'...
Another colleague is also from Togo, and he's double vaxxed.
edit on 27-11-2021 by MonkeyBalls2 because: added stuff
a reply to: angelchemuel
I think that one is quite obvious lol
Someone across the room said "Don't even think about it!"
I think that one is quite obvious lol
Someone across the room said "Don't even think about it!"
POST REMOVED BY STAFF
edit on Sat Nov 27 2021 by DontTreadOnMe because: (no reason given)
I would have been happy with "Nu".
it immediately brought back memories of 2000AD comics, Nu-Earth (Rogue Trooper) and Judge Death (and Fear, Fire and Mortis)....
"Nu Covid" would have been pretty cool
I suppose with Omicron, we have the chance that we can eventually get a Futurama related variant "Omicron Persei 8".
"Laugh in the Face of Death"
it immediately brought back memories of 2000AD comics, Nu-Earth (Rogue Trooper) and Judge Death (and Fear, Fire and Mortis)....
"Nu Covid" would have been pretty cool
I suppose with Omicron, we have the chance that we can eventually get a Futurama related variant "Omicron Persei 8".
"Laugh in the Face of Death"
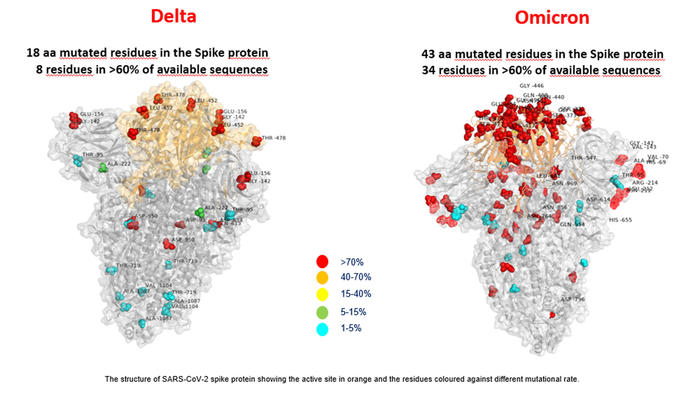
A first glimpse of Omicron compared to Delta :
Originally From ANSA.IT
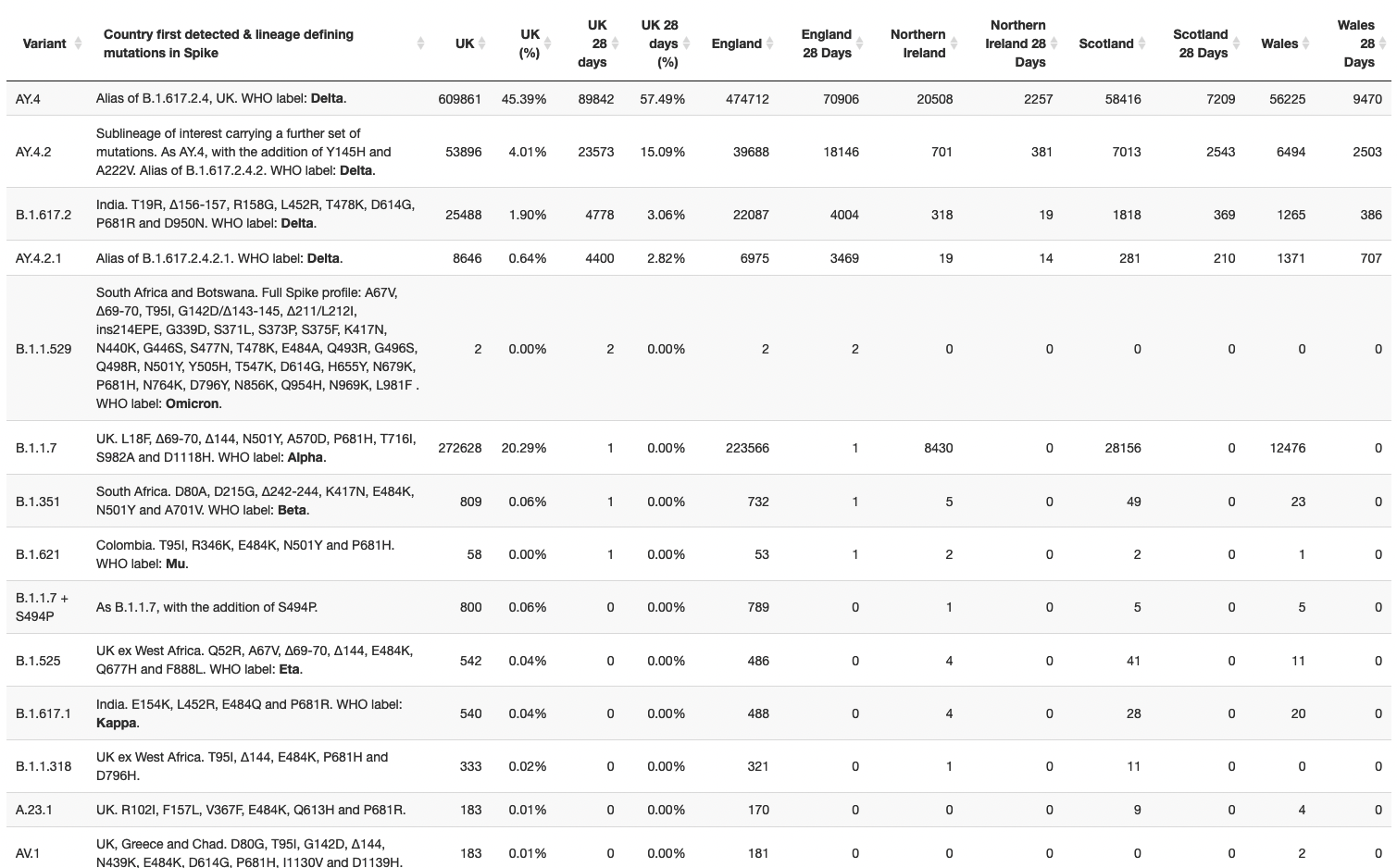
edit : Omicron has also appeared on the COG UK Website takes up a lot of space with all those mutations :
Originally From ANSA.IT

edit : Omicron has also appeared on the COG UK Website takes up a lot of space with all those mutations :

edit on 28-11-2021 by MonkeyBalls2 because: added edit
Later.
edit on 28-11-2021 by Soloprotocol because: (no reason given)
a reply to: Soloprotocol
And none of them related to travel apparently. They are calling it a community infection!
Rainbows
Jane
And none of them related to travel apparently. They are calling it a community infection!
Rainbows
Jane
originally posted by: angelchemuel
a reply to: Soloprotocol
And none of them related to travel apparently. They are calling it a community infection!
Rainbows
Jane
Half of them were 'unvaxxed', apparently.
originally posted by: angelchemuel
a reply to: Soloprotocol
And none of them related to travel apparently. They are calling it a community infection!
Rainbows
Jane
Begs the question. Are the mutations caused by the vaccines?, With different mutations dependent on which vaccine you have in your system?.
edit on 29-11-2021 by Soloprotocol because: (no reason given)
a reply to: Soloprotocol
So was that unvaxx - never jabbed or jabbed and no longer consider fully vaxxed
So was that unvaxx - never jabbed or jabbed and no longer consider fully vaxxed
Can be cited as: Goldstein L., Re-analysis of Molnupiravir Trials, Phase II/III; defyccc.com, PREPRINT, [link], 2021
Abstract
This is a re-analysis of the data from Molnupiravir trials, presented by the FDA.
The trials’ sponsor modified the pre-registered protocol, so the results will appear more beneficial than they are. To this end, results from patients recruited by August 5 were reported as a separate trial – here referred to as Trial 1. Thus, the data from patients recruited after August 5 should also be presented as a separate trial – here referred to as Trial 2.
In this second trial, the Molnupiravir arm had 32.5% higher relative risk of hospitalization or death compared to the placebo arm.
So this seems like a cancel but ?
trial sitenews
a reply to: Soloprotocol
The 'other' various Dr's/scientists that we are 'not allowed' to listen to, have said from the rollout of the vaccine that it will force mutations, now being evidenced. Heck, even the co-inventor of the RNA style vaccine has said it is the wrong type of vaccine for this virus.
Rainbows
Jane
The 'other' various Dr's/scientists that we are 'not allowed' to listen to, have said from the rollout of the vaccine that it will force mutations, now being evidenced. Heck, even the co-inventor of the RNA style vaccine has said it is the wrong type of vaccine for this virus.
Rainbows
Jane
a reply to: angelchemuel
Have you watched this yet?
It's a discussion with Aubrey Marcus and Dr Aditi Bhargavai.
It asks the question....is there enough information and evidence that the vaccine is as safe and effective as advertised?
I have posted it twice already on ATS but you may have missed it...... it's a must watch imo.
Have you watched this yet?
It's a discussion with Aubrey Marcus and Dr Aditi Bhargavai.
It asks the question....is there enough information and evidence that the vaccine is as safe and effective as advertised?
I have posted it twice already on ATS but you may have missed it...... it's a must watch imo.
POST REMOVED BY STAFF
edit on Tue Nov 30 2021 by DontTreadOnMe because: (no reason given)
a reply to: Itisnowagain
That's a long watch! I'll listen to it while I multi task
Thank you.
Rainbows
Jane
That's a long watch! I'll listen to it while I multi task
Thank you.
Rainbows
Jane
new topics
-
Eritrean gangs clash in 10-hour riot during 'cultural celebration' North Carolina
Social Issues and Civil Unrest: 7 minutes ago -
American Taxpayers Fund Hezbollah Terrorist Memorial In Dearborn Michigan
Social Issues and Civil Unrest: 1 hours ago -
Kamala Harris tells Oprah she will kill an intruder
US Political Madness: 3 hours ago -
Illegal Alien brothers arrested in Missouri for connection with deadly carjackings
Social Issues and Civil Unrest: 4 hours ago -
Teenage Girl Raped and Woman Sexually Assaulted on Brighton Beach
Social Issues and Civil Unrest: 6 hours ago -
When locals can spot your counterfeit pandas
Jokes, Puns, & Pranks: 7 hours ago -
Sept 2024 - MATT GAETZ Reveals There Are FIVE Trump Assassination Teams in the USA Right Now.
2024 Elections: 9 hours ago -
BIDEN-HARRIS DOJ-DHS Blocked Florida Law Enforcement from Investigating Would-Be Trump Assassin.
US Political Madness: 10 hours ago -
A music rant hitting ATS politics as well
Rant: 10 hours ago
top topics
-
Ex-Border Patrol Chief Testifies that Biden/Harris Ordered Media Cover Up!
US Political Madness: 15 hours ago, 19 flags -
BIDEN-HARRIS DOJ-DHS Blocked Florida Law Enforcement from Investigating Would-Be Trump Assassin.
US Political Madness: 10 hours ago, 17 flags -
A sheriff has been arrested after shooting at a judge at county courthouse with a fatality
Other Current Events: 15 hours ago, 13 flags -
Oklahoma announced that they have removed 450,000 ineligible names from the voter rolls
2024 Elections: 15 hours ago, 11 flags -
My latest form of art…
Member Art: 12 hours ago, 10 flags -
Kamala Harris tells Oprah she will kill an intruder
US Political Madness: 3 hours ago, 8 flags -
Illegal Alien brothers arrested in Missouri for connection with deadly carjackings
Social Issues and Civil Unrest: 4 hours ago, 8 flags -
Sept 2024 - MATT GAETZ Reveals There Are FIVE Trump Assassination Teams in the USA Right Now.
2024 Elections: 9 hours ago, 7 flags -
Teenage Girl Raped and Woman Sexually Assaulted on Brighton Beach
Social Issues and Civil Unrest: 6 hours ago, 7 flags -
A small potential issue with Kamala.
Politicians & People: 15 hours ago, 4 flags
active topics
-
Ukraine hit 300 miles into Russia
World War Three • 133 • : UKTruth -
Teenage Girl Raped and Woman Sexually Assaulted on Brighton Beach
Social Issues and Civil Unrest • 47 • : SprocketUK -
American Taxpayers Fund Hezbollah Terrorist Memorial In Dearborn Michigan
Social Issues and Civil Unrest • 5 • : watchitburn -
Israel Launches 2nd Wave Of EXPLODING TECH, Hezbollah Radios Begin Exploding
World War Three • 64 • : Lazy88 -
Eritrean gangs clash in 10-hour riot during 'cultural celebration' North Carolina
Social Issues and Civil Unrest • 0 • : watchitburn -
Gavin Newsom passes law against deepfake political ads
Politicians & People • 28 • : strongfp -
Nigel Farage : I am giving up control of the party to its members.
Regional Politics • 10 • : angelchemuel -
My latest form of art…
Member Art • 26 • : Encia22 -
Kamala Harris tells Oprah she will kill an intruder
US Political Madness • 21 • : PorkChop96 -
Ex-Border Patrol Chief Testifies that Biden/Harris Ordered Media Cover Up!
US Political Madness • 22 • : xuenchen
