It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
This is my first time posting a new thread so forgive me if missing or doing something wrong.
Anywho I was just looking around at current news and what not and seen this article about the MonkeyPox/Mpox Vaccine that the FDA approved....And after further looking into it I find it very disturbing that they could even approve of this after seeing ALL the issues that it may cause....I will post links to the articles as well as to the FDA site.
I would like to hear what others think of this info ...Do the Benefits Outweigh the Downsides??
FDA Approves Vaccine for Mpox — Warns It May Cause Death in Vaccinated and People They Come in Contact With - LINK1 - LINK2
FDA MEDICATION GUIDE

FDA News Release: FDA Roundup: August 30, 2024
I would have to say after reading some of the issues from this Vax it's no wonder we have so many anti-vaxxers these days .......But if they are moving forward with more like this .......and the Covid/mRna vax ????
Anywho I was just looking around at current news and what not and seen this article about the MonkeyPox/Mpox Vaccine that the FDA approved....And after further looking into it I find it very disturbing that they could even approve of this after seeing ALL the issues that it may cause....I will post links to the articles as well as to the FDA site.
I would like to hear what others think of this info ...Do the Benefits Outweigh the Downsides??
FDA Approves Vaccine for Mpox — Warns It May Cause Death in Vaccinated and People They Come in Contact With - LINK1 - LINK2
Yes, the ACAM2000 is a live replicating vaccinia virus vaccine that, per the adverse reactions listed on the warnings insert, can cause severe disability, inflammation of the brain and spinal cord, permanent neurological sequelae (long term infection), death of the vaccinated individual, and … wait for it… “death of unvaccinated individuals who have contact with vaccinated individuals.” You cannot make this stuff up.
The CDC website on mpox vaccination states that ACAM2000 “has not been used in the ongoing clade II mpox outbreak that started in 2022″ but “has been made available for use against mpox in the clade II outbreak under an Expanded Access Investigational New Drug (EA-IND) protocol, which requires informed consent along with completing additional forms.”
FDA MEDICATION GUIDE

The guide — which the FDA requires to be given to patients before they get the ACAM2000 vaccine — says, “ACAM2000 can cause serious complications in vaccinated individuals and in their close contacts to whom the vaccine virus has spread.”
FDA News Release: FDA Roundup: August 30, 2024
I would have to say after reading some of the issues from this Vax it's no wonder we have so many anti-vaxxers these days .......But if they are moving forward with more like this .......and the Covid/mRna vax ????
Congratulations on your first post! I always love your replies and how they are backed up with links and personal thoughts on the subjects you
address.
Having that said, I’m going to ask, if before Covid, did traditional vaccines ever sport this kind of warning?
I can’t recall and I’m asking because I’m skeptical by nature. And I don’t wanna muddy the conversation by asking how or in what ways this vaccine is similar to the Covid ones.
Having that said, I’m going to ask, if before Covid, did traditional vaccines ever sport this kind of warning?
I can’t recall and I’m asking because I’m skeptical by nature. And I don’t wanna muddy the conversation by asking how or in what ways this vaccine is similar to the Covid ones.
edit on 24-9-2024 by SteamyAmerican because: (no reason given)
The links work fine for me,
It seems that big pharma money gets into the pockets of politicians to turn their face the other side, that includes the same agencies that are supposed to protect the public.
From the link,
Wow some dangerous vaccine, but hey big pharma does not give a crap who they kill. They got away with the covid jab.
It seems that big pharma money gets into the pockets of politicians to turn their face the other side, that includes the same agencies that are supposed to protect the public.
From the link,
Karl Jablonowski, Ph.D. — senior research scientist at Children’s Health Defense — told The Defender that the FDA’s approval of the shot is a “public hazard, not the practice of public health.”
“You can call ACAM2000 a ‘vaccine,’ but its impact on the United States will be as a ‘disease,’” Jablonowski said.
He added:
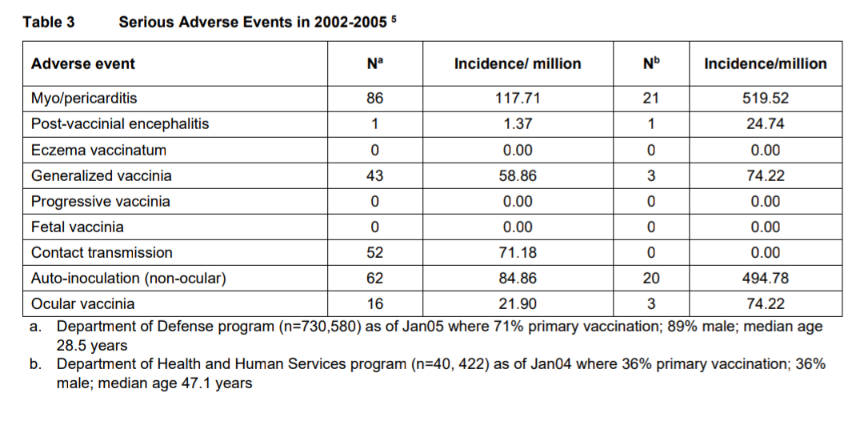
“For instance, the drug’s package insert reveals (see Table 3) that the U.S. Department of Health and Human Services (HHS) found roughly 520 per million ACAM2000 recipients contracted myocarditis or pericarditis.
“That is about 1 in 2,000. But elsewhere in the package insert, the FDA acknowledges that — based on additional studies — the risk is actually 5.7 per 1,000 vaccinations.
“That’s about 1 in every 175 people. If the 262 million adults in the U.S. received this vaccine, an estimated 1,493,000 would contract the serious adverse reaction of myopericarditis.”
Dr. Meryl Nass, an internist and biological warfare expert, told The Defender that the CDC also acknowledged this same figure — myopericarditis in 5.7 per 1,000 primary ACAM2000 vaccinations — during a June 23, 2022, meeting.
“It’s a huge number,” Nass said. “The fact that the FDA and CDC are acknowledging it means no one should get the vaccine.”
She explained:
“Myopericarditis is a life-threatening condition. It lowers your life expectancy.
Wow some dangerous vaccine, but hey big pharma does not give a crap who they kill. They got away with the covid jab.
a reply to: SteamyAmerican
Thank You
I have read up on the MMR vax and it seems to have some issues as well ...but not sure if as bad as this one
I'm sure someone could point out others that might be.....we'll wait and see
Thank You
I have read up on the MMR vax and it seems to have some issues as well ...but not sure if as bad as this one
I'm sure someone could point out others that might be.....we'll wait and see
a reply to: marg6043 Agree 100%.
The fact that they changed the name of the disease, and push this vax while ALSO suppressing that it is generally gay males by which sexual contact infects, AND by and large most of the cases of this disease is pretty telling.
How many times has the FDA pulled a previously authorized vaccine or treatment? Isn’t the Covid shot still under EAU? If so why did they update the schedule for schoolchildren and babies?
Are they giving out free donuts and beer for this new one?
Things to think about.
The fact that they changed the name of the disease, and push this vax while ALSO suppressing that it is generally gay males by which sexual contact infects, AND by and large most of the cases of this disease is pretty telling.
How many times has the FDA pulled a previously authorized vaccine or treatment? Isn’t the Covid shot still under EAU? If so why did they update the schedule for schoolchildren and babies?
Are they giving out free donuts and beer for this new one?
Things to think about.
edit on 24-9-2024 by SteamyAmerican because: (no reason given)
a reply to: tarantulabite1
ACAM2000 is basically a Smallpox vaccine that injects a live Vacinnia virus instead of a dead Smallpox virus. The Smallpox virus and the Mpox virus and the Vacinnia virus are all enough alike that developing immunity to Vacinnia also confers immunity to the other two viruses. Mpox can have a case mortality rate of up to 10%.
By contrast, here's what the prescribing information for ACAM2000 says:
Historically, death following vaccination with live Vacinnia virus is a rare event; approximately 1 death per million primary vaccinations and 1 death per 4 million revaccinations have occurred after vaccination with live Vacinnia virus."
www.fda.gov...#:~:text=adverse%20reactions%20include%20inoculation%20site,%2C%20and%20headache%20(6).
So, 1 in a million chance of dying from a Vacinnia infection (either naturally occurring or from the vaccine) compared to a 1 in 10 chance of dying from a Mpox infection if you haven't been vaccinated.
You decide.
ACAM2000 is basically a Smallpox vaccine that injects a live Vacinnia virus instead of a dead Smallpox virus. The Smallpox virus and the Mpox virus and the Vacinnia virus are all enough alike that developing immunity to Vacinnia also confers immunity to the other two viruses. Mpox can have a case mortality rate of up to 10%.
By contrast, here's what the prescribing information for ACAM2000 says:
Historically, death following vaccination with live Vacinnia virus is a rare event; approximately 1 death per million primary vaccinations and 1 death per 4 million revaccinations have occurred after vaccination with live Vacinnia virus."
www.fda.gov...#:~:text=adverse%20reactions%20include%20inoculation%20site,%2C%20and%20headache%20(6).
So, 1 in a million chance of dying from a Vacinnia infection (either naturally occurring or from the vaccine) compared to a 1 in 10 chance of dying from a Mpox infection if you haven't been vaccinated.
You decide.
Boy have they flipped the tables! Instead of the unvaccinated being hazardous to be around, it looks like these 'vaccines' will make the vaccinated
deadly to those around them for 6 weeks or so, if they happen to be UNVACCINATED that is. So is this a new twist on encouraging getting this
vaccine?
Becoming a hermit is looking more and more appealing.
Becoming a hermit is looking more and more appealing.
a reply to: Boomer1947
From FDA
Articale
From FDA
“For instance, the drug’s package insert reveals (see Table 3) that the U.S. Department of Health and Human Services (HHS) found roughly 520 per million ACAM2000 recipients contracted myocarditis or pericarditis.
“That is about 1 in 2,000. But elsewhere in the package insert, the FDA acknowledges that — based on additional studies — the risk is actually 5.7 per 1,000 vaccinations.
“That’s about 1 in every 175 people. If the 262 million adults in the U.S. received this vaccine, an estimated 1,493,000 would contract the serious adverse reaction of myopericarditis.”
Articale
“Myopericarditis is a life-threatening condition. It lowers your life expectancy.
“Most people are never going to be exposed to mpox or smallpox. Vaccines are typically given to healthy people who are never even going to be exposed, therefore the risk posed by the vaccine needs to be low.
“That’s why vaccine safety is critical — even more than drugs, because drugs are given to people who are already sick.
“This risk — 1 in 175 — is higher than all other licensed vaccines for which the CDC acknowledges side effects. This is the worst.”
edit on 24-9-2024 by tarantulabite1 because: (no reason given)
They're setting up a new "MASK/QUARANTEEN" scenario at the will of Government Mandate. ⚔️☠️
😉🤔

😉🤔

a reply to: tarantulabite1
Those warnings are merely stepped-up labels to doubly protect big pharms in case things don't work out. They are "running gun-shy" you know.
Those warnings are merely stepped-up labels to doubly protect big pharms in case things don't work out. They are "running gun-shy" you know.
a reply to: SteamyAmerican
Soon will be pushed to be mandatory for babies and children, that is how big pharma makes sure their jabs are protected.
They tried with covid jab until individual states make their own rules about it.
Interesting to point out that it seems, those receiving the jab can infect those around them, meaning it can spread the disease fast.
Sound like another pandemic via jabbed people what a way to increase contagion.
Soon will be pushed to be mandatory for babies and children, that is how big pharma makes sure their jabs are protected.
They tried with covid jab until individual states make their own rules about it.
Interesting to point out that it seems, those receiving the jab can infect those around them, meaning it can spread the disease fast.
Sound like another pandemic via jabbed people what a way to increase contagion.
edit on 24-9-2024 by marg6043 because: (no reason given)
originally posted by: Boomer1947
a reply to: tarantulabite1
So, 1 in a million chance of dying from a Vacinnia infection (either naturally occurring or from the vaccine) compared to a 1 in 10 chance of dying from a Mpox infection if you haven't been vaccinated.
You decide.
I think most of us have it under control since we don't have gay sex with multiple strangers on a routine basis.
As I pointed out before, this is the worst public forum in the world for psuedo-intellectuals with a pharma fetish to be posting on. Virtually nowhere else would allow me, or anybody else that actually knows the topic, to speak at length on all this pharma marketing and globalist alarmism you constantly peddle.
Although many reports state that 10% of clade I infections in humans are fatal, infectious-disease researcher Laurens Liesenborghs at the Institute of Tropical Medicine in Antwerp, Belgium, doubts that this figure is accurate. Even the WHO’s latest estimate of a 3.5% fatality rate for people with mpox in the DRC might be high.
There are many reasons that fatality estimates might be unreliable, Liesenborghs says. For one, surveillance data capture only the most severe cases; many people who are less ill might not seek care at hospitals or through physicians, so their infections go unreported.
Another factor that can confound fatality rates is a secondary health condition. For example, people living with HIV — who can represent a large proportion of the population in many African countries — die from mpox at twice the rate of the general population2, especially if their HIV is untreated. And the relatively high death rate among children under age 5 could be partly because of malnutrition, which is common among kids in rural parts of the DRC, Liesenborghs says.
Nature
In first world countries Mpox is an STD found mostly in promiscuous men that have sex with men.
Up to 22 June 2022, 508 confirmed cases of monkeypox (MPX) have been reported in the Madrid region of Spain, 99% are men (n = 503) with a median age of 35 years (range: 18–67). In this ongoing outbreak, 427 cases (84.1%) reported condomless sex or sex with multiple partners within the 21 days before onset of symptoms, who were predominantly men who have sex with men (MSM) (n = 397; 93%). Both the location of the rash, mainly in the anogenital and perineal area, as well as the presence of inguinal lymphadenopathy suggest that close physical contact during sexual activity played a key role in transmission.
The Science©



Vaccine insert
Over 90% had some adverse reaction and 10% had a serious one.
Everyone please remember the following when they start pushing it on kids and memory hole these CDC recommendations.
Get the vaccine if you:
Are a gay, bisexual, or other same-gender loving man who has sex with men or are transgender, gender non-binary, or gender-diverse AND in the last 6 months you have had, or expect to have
One or more sexually transmitted infections
More than one sexual partner, or anonymous sexual or intimate contact
Sex at a commercial sex venue
Sex in association with a large public event in a geographic area where mpox transmission is occurring
(post by underpass61 removed for a manners violation)
Can the vaccine itself mutate in the recipient before being passed onto to others?
If so then this is an even worse idea than it already appeared to be at first glance.
If so then this is an even worse idea than it already appeared to be at first glance.
a reply to: tarantulabite1
You may find this article published by Brownstone author Sonia Elijah of interest. She has been following the mRNA flavored jabs and now has concerns about this Mpox jab.
Mpox, Project Bioshield: Everyone's Dark Winter.
I have wondered why the name was changed from Monkeypox. Nothing racists or sexists about it.
ON in numbers...
O=15
N=11...'26. The next plandemic is going to be hot in 2026.
KE
K=14
E=5...'19. The start of the Covid plandemic.
You may find this article published by Brownstone author Sonia Elijah of interest. She has been following the mRNA flavored jabs and now has concerns about this Mpox jab.
Mpox, Project Bioshield: Everyone's Dark Winter.
I have wondered why the name was changed from Monkeypox. Nothing racists or sexists about it.
ON in numbers...
O=15
N=11...'26. The next plandemic is going to be hot in 2026.
KE
K=14
E=5...'19. The start of the Covid plandemic.
originally posted by: McGinty
Can the vaccine itself mutate in the recipient before being passed onto to others?
If so then this is an even worse idea than it already appeared to be at first glance.
You'll find the post vaccination precaution for monkey pox is pretty much the same as for small pox vaccine. It's a live vaccinia virus. Same as small pox. Anyone susceptible to the vaccinia virus can potentially get vaccinia related illness when exposed to the vaccine site or anything that has contacted it.
Remember, the small pox vaccine (which is pretty much the same used for monkey pox) was so highly effective (unlike one popularized in 2020 timeframe) it eliminated small pox in the wild. So effective it was no longer given because the bug was no longer naturally occuring in the world. It was brought back for military use, I forget when, due to "perceived biological warfare threat", but never brought back as a public recommendation as the threat is so low.
I'm in my mid 50's and never got the vaccine as a child. Those younger may not even know about the vaccine or how effective it is, or that post vaccination precautions associated with it were accepted as no big deal. Ya followed them. Ya stayed away from susceptible people until the scab fell off and moved on.
The whole C19 vax fiasco caused a lot of people to lose faith in vaccines in general, which is a bit sad, because the oldies, but goodies were incredibly effective in actually preventing illness and spread of disease... the small pox vaccine being the most effective vaccine program ever.
new topics
-
Meta Llama local AI system is scary good
Science & Technology: 2 hours ago -
This is why ALL illegals who live in the US must go
Social Issues and Civil Unrest: 4 hours ago -
UK Borders are NOT Secure!
Social Issues and Civil Unrest: 5 hours ago -
Former ‘GMA Producer’ Sues NPR-Legacy Media Exposed
Propaganda Mill: 7 hours ago -
New Footage - Randy Rhoads 1979 LIVE Guitar Solo Footage at the Whisky - Pro Shot
Music: 8 hours ago -
Happy Hanukkah…
General Chit Chat: 8 hours ago
top topics
-
This is why ALL illegals who live in the US must go
Social Issues and Civil Unrest: 4 hours ago, 11 flags -
Former ‘GMA Producer’ Sues NPR-Legacy Media Exposed
Propaganda Mill: 7 hours ago, 8 flags -
A Merry Christmas.
General Chit Chat: 15 hours ago, 7 flags -
Happy Hanukkah…
General Chit Chat: 8 hours ago, 7 flags -
UK Borders are NOT Secure!
Social Issues and Civil Unrest: 5 hours ago, 6 flags -
New Footage - Randy Rhoads 1979 LIVE Guitar Solo Footage at the Whisky - Pro Shot
Music: 8 hours ago, 5 flags -
Meta Llama local AI system is scary good
Science & Technology: 2 hours ago, 4 flags
active topics
-
Orange County Makes Shoplifting a Felony
Other Current Events • 31 • : GENERAL EYES -
This is why ALL illegals who live in the US must go
Social Issues and Civil Unrest • 9 • : Flyingclaydisk -
Meta Llama local AI system is scary good
Science & Technology • 10 • : glend -
Happy Hanukkah…
General Chit Chat • 20 • : JJproductions -
NYPD arrests migrant who allegedly set woman on fire on subway train, watched her burn to death
Breaking Alternative News • 55 • : xuenchen -
Plane Crash Today --Azerbaijanian E190 passenger jet
Mainstream News • 36 • : Oldcarpy2 -
FAA Investigates Christmas Drone Show Gone Wrong in Orlando, FL 12/2024
Other Current Events • 11 • : ScottKirkland -
It's Offical Now
US Political Madness • 26 • : Oldcarpy2 -
This is an interesting picture. Do we actually pick our leaders?
Politicians & People • 13 • : GENERAL EYES -
Panamanian President-“every square meter” of the Panama Canal belongs to Panama.
New World Order • 45 • : SCUBAPAUL
