It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
Hi, I’m John Skieswanne, and this post is part 1 of a series on physics. In this series I will explain a few pillars of modern physics. I won't be
using any complex maths. It is my hope that this series will introduce some of you brilliant, curious-minded laymen out there to the inner circles of
Physics.
So, sit back and enjoy.
******
the Uncertainty Principle.
In the quantum model, once you reach the subatomic level, nothing is certain anymore. In fact, one of the major pillars of the Quantum Model (QM, for short) is actually named the "Uncertainty Principle".
When we measure the position of "large" objects, their position doesn't get much disturbed. This applies for galaxies, for stellar objects, and for small objects such as watches and T-shirts and tennis balls. This even works for most atoms. But one thing Werner Heisenberg discovered in 1927: there is a lower limit to this process. Past a certain point, there is no way to know for sure where a subatomic particle is located anymore. At such a small scale, we can only say these particles have a probable position.
Here's an analogy to illustrate Heisenberg's argument:
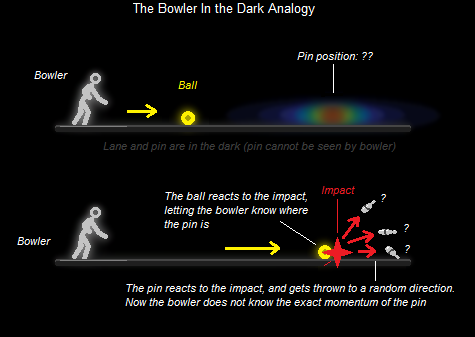
Imagine an electron (the target particle) as a lonely pin on a very, very dark bowling lane. You can't see the pin, you can only guess where it is. Since you don't know yet the position of the pin on the lane, you may think of the pin as being smudged all over the lane as a probability amplitude - meaning it can be nearly everywhere inside this wave-like function of possibilities, but at some places more likely than others:

Heisenberg pointed out that to figure out where the electron (our bowling pin) is, you have to send some kind of signal onto it, like a light or something (you can't know its position if you don't detect it in the first place).
Now light rays are made of particles, called photons.
Imagine you have a bowling ball - a glow-in-the-dark bowling ball. This ball represents the photon - the particle of light. Now imagine you throw the bowling ball onto the lane, at the pin somewhere in the dark. Luckily, you hit the pin with the very first shot - and at the point of impact, you see your bowling ball deviate. Now, you know where the pin is, right? - It is located at the point where your bowling ball got deviated.
Think again.
Because of the impact itself, the pin has been kicked back at some other, random direction. Thus although its position may be known at the moment of impact, its momentum (and momentum direction) is not known with certainty anymore.
******
Thus, Heisenberg concluded that at the subatomic level, no machines can accurately pinpoint the exact position of a particle without compromising its momentum, and vice-versa. That's basically because the target particles are so small, that any measurements, such as those performed by sending a photon at them, would upset their state (by distributing energy to them).
That is why in the Quantum Model the properties (such as position, momentum, etc) of a particle are represented using probabilities. Since we just don't know, we assign this probability wave function to a particle. Once you know one property of the particle for certain, then you may say that you "collapsed the particle's wave function" - this basically means that you've eliminated all other possibilities but one (for this specific property) down to zero.
A common mistake, especially in New Age, is to confuse "subatomic measurement" with "human eye". These are two different kind of "observations". In physics, "observation" really means "measurement" - not necessarily organic, visual or spiritual, but actually physical.
You can stare in the dark for eons - this will not change the wave functions of the particles there. To collapse the wave function you actually have to send something at it, you have to interfere with it in some way, otherwise it can't "know" if you're measuring it or not in the first place. Additionally, Heisenberg's Uncertainty vanishes astronomically as soon as the target object becomes large enough to sustain measurement without getting kicked to some random direction. So far, galaxies, planets and even humans do not receive that much of a kick from the Reception of One Photon. Accordingly, these larger objects are dominated by very tangible laws ("classical" laws of physics) discovered by Newton and Einstein ages ago.
******
I hope you enjoyed this read; Part 2 will be coming soon, and will deal about Einstein's Special Relativity - a very popular aspect of modern physics.
Swan
So, sit back and enjoy.
******
the Uncertainty Principle.
In the quantum model, once you reach the subatomic level, nothing is certain anymore. In fact, one of the major pillars of the Quantum Model (QM, for short) is actually named the "Uncertainty Principle".
When we measure the position of "large" objects, their position doesn't get much disturbed. This applies for galaxies, for stellar objects, and for small objects such as watches and T-shirts and tennis balls. This even works for most atoms. But one thing Werner Heisenberg discovered in 1927: there is a lower limit to this process. Past a certain point, there is no way to know for sure where a subatomic particle is located anymore. At such a small scale, we can only say these particles have a probable position.
Here's an analogy to illustrate Heisenberg's argument:
Imagine an electron (the target particle) as a lonely pin on a very, very dark bowling lane. You can't see the pin, you can only guess where it is. Since you don't know yet the position of the pin on the lane, you may think of the pin as being smudged all over the lane as a probability amplitude - meaning it can be nearly everywhere inside this wave-like function of possibilities, but at some places more likely than others:

Heisenberg pointed out that to figure out where the electron (our bowling pin) is, you have to send some kind of signal onto it, like a light or something (you can't know its position if you don't detect it in the first place).
Now light rays are made of particles, called photons.
Imagine you have a bowling ball - a glow-in-the-dark bowling ball. This ball represents the photon - the particle of light. Now imagine you throw the bowling ball onto the lane, at the pin somewhere in the dark. Luckily, you hit the pin with the very first shot - and at the point of impact, you see your bowling ball deviate. Now, you know where the pin is, right? - It is located at the point where your bowling ball got deviated.
Think again.
Because of the impact itself, the pin has been kicked back at some other, random direction. Thus although its position may be known at the moment of impact, its momentum (and momentum direction) is not known with certainty anymore.

******
Thus, Heisenberg concluded that at the subatomic level, no machines can accurately pinpoint the exact position of a particle without compromising its momentum, and vice-versa. That's basically because the target particles are so small, that any measurements, such as those performed by sending a photon at them, would upset their state (by distributing energy to them).
That is why in the Quantum Model the properties (such as position, momentum, etc) of a particle are represented using probabilities. Since we just don't know, we assign this probability wave function to a particle. Once you know one property of the particle for certain, then you may say that you "collapsed the particle's wave function" - this basically means that you've eliminated all other possibilities but one (for this specific property) down to zero.
A common mistake, especially in New Age, is to confuse "subatomic measurement" with "human eye". These are two different kind of "observations". In physics, "observation" really means "measurement" - not necessarily organic, visual or spiritual, but actually physical.

You can stare in the dark for eons - this will not change the wave functions of the particles there. To collapse the wave function you actually have to send something at it, you have to interfere with it in some way, otherwise it can't "know" if you're measuring it or not in the first place. Additionally, Heisenberg's Uncertainty vanishes astronomically as soon as the target object becomes large enough to sustain measurement without getting kicked to some random direction. So far, galaxies, planets and even humans do not receive that much of a kick from the Reception of One Photon. Accordingly, these larger objects are dominated by very tangible laws ("classical" laws of physics) discovered by Newton and Einstein ages ago.
******
I hope you enjoyed this read; Part 2 will be coming soon, and will deal about Einstein's Special Relativity - a very popular aspect of modern physics.
Swan
a reply to: swanne
That's a great thread idea, and you have used decent analogies to explain the Uncertainty Principle. I think that it is great that you are intending to illuminate physics for the less mathematically minded. I for one am useless at mathematics, but found that through research and the watching of many documentaries, I was able to build up a decent understanding of these things.
I applaud you for taking on the task of normalising these concepts so that they can be assimilated by the layperson!
Good job!
That's a great thread idea, and you have used decent analogies to explain the Uncertainty Principle. I think that it is great that you are intending to illuminate physics for the less mathematically minded. I for one am useless at mathematics, but found that through research and the watching of many documentaries, I was able to build up a decent understanding of these things.
I applaud you for taking on the task of normalising these concepts so that they can be assimilated by the layperson!
Good job!
S&F Nice breakdown, i would like to see more of these posts.
Keep up the good work
Luke
Keep up the good work
Luke
a reply to: swanne
I recall reading another discussion you were having quite some time ago. The only thing I definitely remember about it was that I could understand one word in fifty, and can't recall the subject. Thanks for stepping things down.
My understanding of Heisenberg's Principle remains unchanged however.
"If we mess with something, we won't be able to fix it."
I recall reading another discussion you were having quite some time ago. The only thing I definitely remember about it was that I could understand one word in fifty, and can't recall the subject. Thanks for stepping things down.
My understanding of Heisenberg's Principle remains unchanged however.
"If we mess with something, we won't be able to fix it."
Have you ever read Robert Anton Wilson's 'Shroedinger's Cat'? It applies the Uncertainty Principle to the macro-scale. Makes for a damn
entertaining read.
a reply to: skunkape23
Indeed.
I personally favour the Many-World interpretation - in my humble opinion it is the only way to solve the so-called time travel paradox.
Indeed.
I personally favour the Many-World interpretation - in my humble opinion it is the only way to solve the so-called time travel paradox.
a reply to: charles1952
Well, if I may, it's more like,
"If we mess with a mess, it stays a mess. "
The World is no perfect place... but then it'd be boring if it was perfect.
Well, if I may, it's more like,
"If we mess with a mess, it stays a mess. "
The World is no perfect place... but then it'd be boring if it was perfect.
originally posted by: skunkape23
Have you ever read Robert Anton Wilson's 'Shroedinger's Cat'? It applies the Uncertainty Principle to the macro-scale. Makes for a damn entertaining read.
I will second this motion, and agree that it's an DER. Wilson explains things in simple terms as well, which is the core of this single volume (made up of a trilogy of earlier books) and his other works.
The part 2 is a bit more challenging to write so it may take a while. Meanwhile, if you guys have any questions regarding Heisenberg's Uncertainty
Principle, fire away.
Then how does a quantum computer work? I've heard it is based entirely on the Uncertainty principle, but how can they make it work?
Ok, so here's a question. Using the colored image of the location of a particle. There's an area in the center where it's most likely to be, with
decreasing probability as it moves out from there. Lets say you have a million particles like this and you begin measuring them to see if the
particle is in a specific area, starting at the lowest probability areas and moving inward. Eventually you're going to end up with a situation where
you'll have discovered the location of most particles however some of them still won't have had the location of the particle measured. Through the
process of elimination however you're going to know with certainty where they are. This gives you a group where you know the location of something
without having actually measured and verified it.
How does this not violate Heisenburg?
How does this not violate Heisenburg?
a reply to: Aazadan
To know with certainty where a particle is, you must measure it so to be sure it is indeed there. And although this could indeed give you a big clue as to where all the other particles are packed up, Heisenberg's Uncertainty stays effective, because you still don't know (with 100% certitude) the momentum (and other properties, such as spin direction) of all the particles you did not measured.
Think of it like a randomly-shuffled deck of cards (face down). Although you do know where all 52 cards are (they are under the top-most card), you don't know yet which one is a diamond or which one is a King without flipping them individually.
So, the uncertainty is not violated.
Through the process of elimination however you're going to know with certainty where they are. This gives you a group where you know the location of something without having actually measured and verified it.
How does this not violate Heisenburg?
To know with certainty where a particle is, you must measure it so to be sure it is indeed there. And although this could indeed give you a big clue as to where all the other particles are packed up, Heisenberg's Uncertainty stays effective, because you still don't know (with 100% certitude) the momentum (and other properties, such as spin direction) of all the particles you did not measured.
Think of it like a randomly-shuffled deck of cards (face down). Although you do know where all 52 cards are (they are under the top-most card), you don't know yet which one is a diamond or which one is a King without flipping them individually.
So, the uncertainty is not violated.
I think this is the first time that I've had something in physics explained to me and I got it the first time without going, "huh?"
I have a basic understanding of physics for my job, things such as Newton's law and Boyle's law. The rest is, well, too difficult for a simple labor worker such as myself. lol
I have a basic understanding of physics for my job, things such as Newton's law and Boyle's law. The rest is, well, too difficult for a simple labor worker such as myself. lol
originally posted by: starheart
Then how does a quantum computer work? I've heard it is based entirely on the Uncertainty principle, but how can they make it work?
I don't know all the details but basically a qubit (quantum bit) contains the values 0 and 1 simultaneously. Contrast this with a regular bit which is either 0 or 1. Because of this, an arrangement of qubits are in all possible machine states at all times. From there it uses quantum mechanics that are way above my head to determine the probability of any given machine state being the desired one. From that point you can take the high probability machine states in order to get the information you want.
This allows certain types of computational problems to be solved very quickly (breaking encryption for example). But it's also rather poor for other problems.
new topics
-
2nd Day Thanksgiving!...(leftovers!!)
General Chit Chat: 3 minutes ago -
New Disney Star Wars Films Failing Test of Time?
Movies: 11 hours ago
top topics
-
New Disney Star Wars Films Failing Test of Time?
Movies: 11 hours ago, 5 flags -
The Cost of True Discipleship—Count the Cost
Religion, Faith, And Theology: 16 hours ago, 1 flags -
2nd Day Thanksgiving!...(leftovers!!)
General Chit Chat: 3 minutes ago, 0 flags
active topics
-
The Party of Peace - Trump Cabinet Picks Targeted with Death Threats
US Political Madness • 50 • : xuenchen -
2nd Day Thanksgiving!...(leftovers!!)
General Chit Chat • 0 • : Flyingclaydisk -
Unidentified Flying Objects Over U.S. Military Bases in Northeast UK, as of roughly 11 a.m. CST.
Aliens and UFOs • 31 • : ARM19688 -
Results of the use of the Oreshnik missile system in Dnepropetrovsk
World War Three • 263 • : DontTreadOnMe -
Anyone like the Scorpions?
Music • 23 • : Flyingclaydisk -
New Disney Star Wars Films Failing Test of Time?
Movies • 16 • : Tolkien -
The Cost of True Discipleship—Count the Cost
Religion, Faith, And Theology • 5 • : alwaysbeenhere2 -
-@TH3WH17ERABB17- -Q- ---TIME TO SHOW THE WORLD--- -Part- --44--
Dissecting Disinformation • 3415 • : AianawaQ1320 -
Mass UAP events. DC. Machester Airport, UFOs over sub base in CT, Nuke bases.
Aliens and UFOs • 24 • : devilhunter69 -
Are there any good shows
Television • 69 • : Coelacanth55
