It looks like you're using an Ad Blocker.
Please white-list or disable AboveTopSecret.com in your ad-blocking tool.
Thank you.
Some features of ATS will be disabled while you continue to use an ad-blocker.
share:
A pill has cut the risk of hospitalization or death from Covid-19 by half in a study, Merck and Ridgeback Biotherapeutics said Friday.
Yep we really never needed a jab, that is not a vaccine per say and that does not cure or stop covid and is not 100% effective, you still will get covid and died from the virus.
In my opinion we were lie too, miss guided and manipulated into taking a jab that is not really necessary
The covid scamongering house of card and the fascism that has taken over the country will be put on test with this pill.
Let see how the Dictators in big Pfizer will digest this pill now when people will have choices.
www.msn.com...
I always wonder the maniac ways that the jab has been pushed on people. You can not keep wondering what really is in the darn jab.
Yep we really never needed a jab, that is not a vaccine per say and that does not cure or stop covid and is not 100% effective, you still will get covid and died from the virus.
In my opinion we were lie too, miss guided and manipulated into taking a jab that is not really necessary
At the interim analysis, molnupiravir reduced the risk of hospitalization or death by approximately 50%," Merck said in a news release. "7.3% of patients who received molnupiravir were either hospitalized or died through Day 29 following randomization (28/385), compared with 14.1% of placebo treated patients (53,377). Through Day 29, no deaths were reported in patients who received molnupiravir, as compared to 8 deaths in patients who received placebo."
The covid scamongering house of card and the fascism that has taken over the country will be put on test with this pill.
Let see how the Dictators in big Pfizer will digest this pill now when people will have choices.
www.msn.com...
I always wonder the maniac ways that the jab has been pushed on people. You can not keep wondering what really is in the darn jab.
So far my healthy immune system has protected me. Vitamin D and Zinc and sunshine works for me.
I'm going to pass on any products from Pharma to 'protect' me from a virus with a 99% survival rate.
I'm going to pass on any products from Pharma to 'protect' me from a virus with a 99% survival rate.
Ivermectin and HCQ in varying different protocols from multiple different groups of doctors all across the world has shown that it works, and it works
well.
You can't patent those drugs though, so big pharma cannot make billions in taxpayer money for them.
You can't patent those drugs though, so big pharma cannot make billions in taxpayer money for them.
Was this study funded by Merck? Yeah, no thanks. Why are people still trusting the corrupt, money-hungry drug companies?🤦🏼♀️
This only shows one thing, at least for me, that the jabs were no necessary and that covid can be treated just like you treat any seasonal flu or
upper respiratory infections, the biggest scam in history and we now see it for what it was.
A pill that can treat covid, just think about it, why then the needed to inject the entire world population with a experimental drug, in a way that borders on maniac intent.
That is the part that bothers me the most. What is the real agenda behind the jabs.
A pill that can treat covid, just think about it, why then the needed to inject the entire world population with a experimental drug, in a way that borders on maniac intent.
That is the part that bothers me the most. What is the real agenda behind the jabs.
And when the FDA approves it for use in the US
Biden will restrict it from being delivered to Republican states
Just like he has restricted the latest lifesaving COVID drug from being delivered to Republican states
www.politico.com...
He said he would get revenge on Governors who don't go along with his every whim, and he has carried it out by restricting a lifesaving drug from going to Republican states, he'll do the same with this new drug, I am positive.
That is the way Democrats work these days.
Don't agree with Democrats/liberals/progressives 100%,
you deserve to die,
and now they are carrying it out
by restricting a lifesaving drug from being delivered to Republicans.
Biden will restrict it from being delivered to Republican states
Just like he has restricted the latest lifesaving COVID drug from being delivered to Republican states
www.politico.com...
He said he would get revenge on Governors who don't go along with his every whim, and he has carried it out by restricting a lifesaving drug from going to Republican states, he'll do the same with this new drug, I am positive.
That is the way Democrats work these days.
Don't agree with Democrats/liberals/progressives 100%,
you deserve to die,
and now they are carrying it out
by restricting a lifesaving drug from being delivered to Republicans.
edit on 10/1/21 by The2Billies because: formatting grammar
Have I beaten the odds for not dying? What's more rare a normal person not dying from covid, or a covid-19 shot recipient dying from the virus? What
percentage of people have been overlooked because of the priority to covid?
originally posted by: MaplePatriot
Ivermectin and HCQ in varying different protocols from multiple different groups of doctors all across the world has shown that it works, and it works well.
You can't patent those drugs though, so big pharma cannot make billions in taxpayer money for them.
More importantly, Donald Trump made the suggestion of treating coronvirus with that medicine, so democrats fcked the entire population in order to gain power.
LOL!
So y'all are okay with this experimental drug?
Is it time for me to switch sides of the argument to point all your hypocrisy. Hahaha
So y'all are okay with this experimental drug?
Is it time for me to switch sides of the argument to point all your hypocrisy. Hahaha
originally posted by: LSU2018
originally posted by: MaplePatriot
Ivermectin and HCQ in varying different protocols from multiple different groups of doctors all across the world has shown that it works, and it works well.
You can't patent those drugs though, so big pharma cannot make billions in taxpayer money for them.
More importantly, Donald Trump made the suggestion of treating coronvirus with that medicine, so democrats fcked the entire population in order to gain power.
Yep by that time, they realized COVID was the opportunity and they LIED to all of us. Hell as a 57 year old man I knew TPTB would lie to us. The funny thing is the younger generation who usually has total disdain for anything governmental, bought this chit hook line and sinker....
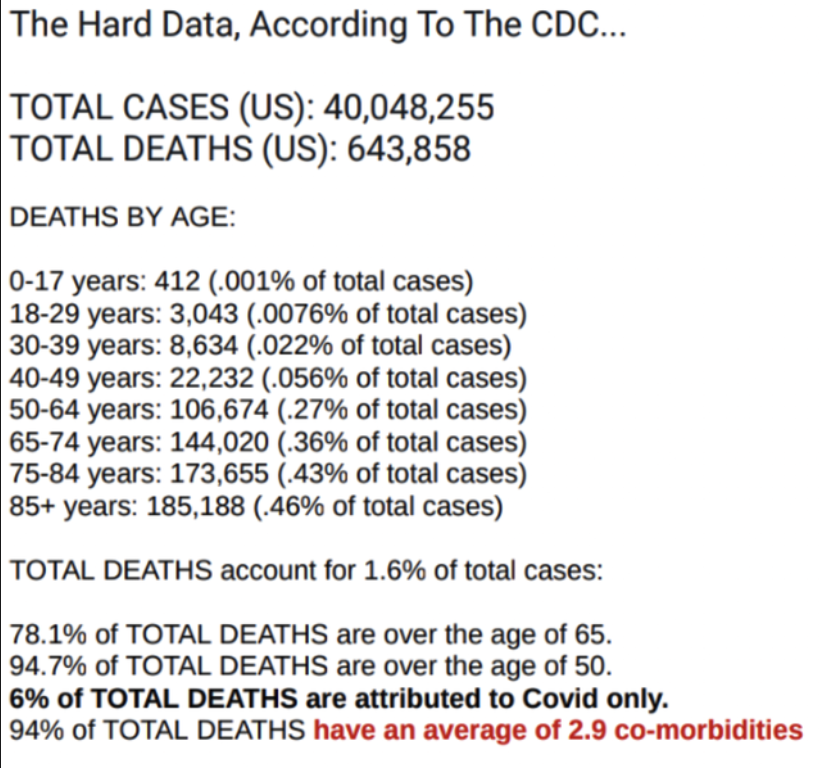
a reply to: putnam6
The zombie generation were the ones that ate the crap like it was candy and the hype that they were going to die, if they did not took the jab.
Hopefully parents will see this, grow a brain and put one and one together.
Covid was the biggest scam in human history and the house of cards are tumbling down, but the darn long term side effects of the jab is going to be for ever and is not way to reverse it.
And is just starting. I predict, that the boosters will bring another wave of infections within the ones that got the jab and covid will be back, take the jab or die
The zombie generation were the ones that ate the crap like it was candy and the hype that they were going to die, if they did not took the jab.
Hopefully parents will see this, grow a brain and put one and one together.
Covid was the biggest scam in human history and the house of cards are tumbling down, but the darn long term side effects of the jab is going to be for ever and is not way to reverse it.
And is just starting. I predict, that the boosters will bring another wave of infections within the ones that got the jab and covid will be back, take the jab or die
edit on 1-10-2021 by marg6043 because: (no reason given)
I COMPLETELY AGREE! #METOO. can you guess what's in this little magic pill? ivermectin folks. repackaged to sell you a more expensive option! works
exactly the same. i wonder hoe much all of this compliance is putting into the pockets of the medical machine? need a covid test to attend a concert,
sporting event, travel etc etc etc... taxation folks... without the tax!
originally posted by: Salander
So far my healthy immune system has protected me. Vitamin D and Zinc and sunshine works for me.
I'm going to pass on any products from Pharma to 'protect' me from a virus with a 99% survival rate.
Yep Merck the makers of ivermectin got a gold mine in their hands for years, now they just renamed and now if approved they will be making billions
and trillions and no need for a jab.
Ivermectin is no an expensive medication but I am sure the new pill will be.
Ivermectin is no an expensive medication but I am sure the new pill will be.
edit on 1-10-2021 by marg6043 because: (no reason
given)
originally posted by: MDDoxs
LOL!
So y'all are okay with this experimental drug?
Is it time for me to switch sides of the argument to point all your hypocrisy. Hahaha
You really don't know the difference between an mRNA injection you are forced to take whether sick or not verse a medicinal pill you take if you get sick?
a reply to: marg6043
They just keep bringing out the scary stuff....
So, get a jab that can alter your system, destroy your immunity via ADE or maybe create altered genetic states OR get a pill that is mutagenic that hasn't even had animal trials yet lol.
Kinda sh!t choices according to the narrative eh?
Or take hydroxychloroquine/quinine or Ivermectin and some vitamins, hmmmmm....
Cheers - Dave
They just keep bringing out the scary stuff....
Safety controversy
In April 2020, Rick Bright, who was removed as head of the Biomedical Advanced Research and Development Authority (BARDA) before the approval of the drug, submitted a whistleblower complaint asserting that Ridgeback pressured BARDA to provide funding to manufacture EIDD-2801 despite Bright's concerns that similar drugs in its class have mutagenic properties.[4]
A previous company, Pharmasset, that had investigated the drug's active ingredient had abandoned it over similar concerns.[5] These claims were denied by George Painter, CEO of DRIVE, noting that toxicity studies on molnupiravir had been carried out and data provided to regulators in the US and UK, who permitted safety studies in humans to move forward in the spring of 2020. Also at this time, DRIVE and Ridgeback Biotherapeutics stated they planned future safety studies in animals.[6]
So, get a jab that can alter your system, destroy your immunity via ADE or maybe create altered genetic states OR get a pill that is mutagenic that hasn't even had animal trials yet lol.
Kinda sh!t choices according to the narrative eh?
Or take hydroxychloroquine/quinine or Ivermectin and some vitamins, hmmmmm....
Cheers - Dave
new topics
-
Leading Surgeon from Al-Shifa Hospital Dies in Israeli Custody
Middle East Issues: 51 minutes ago -
Zionists of ATS assemble
Political Issues: 4 hours ago -
For Votes - President BIDEN Opens ObamaCare Health Ins to Illegal Aliens Eff Nov 1st 2024.
2024 Elections: 9 hours ago -
EPA sues San Francisco for dumping billions of gallons of sewage into Pacific Ocean
US Political Madness: 10 hours ago -
Prophets versus priests; - Getting God's help
Religion, Faith, And Theology: 11 hours ago
top topics
-
Wounded Orangutan seen using a Plant as Medicine for the First Time
Fragile Earth: 15 hours ago, 17 flags -
Study Finds The More Covid Vaccine Doses You Have, the More Likely You Are to Get Covid
Medical Issues & Conspiracies: 14 hours ago, 14 flags -
Farmers jailed without due process
General Conspiracies: 16 hours ago, 10 flags -
EPA sues San Francisco for dumping billions of gallons of sewage into Pacific Ocean
US Political Madness: 10 hours ago, 9 flags -
For Votes - President BIDEN Opens ObamaCare Health Ins to Illegal Aliens Eff Nov 1st 2024.
2024 Elections: 9 hours ago, 8 flags -
UN Estimates Rebuilding Gaza Will Cost Up To 40 Billion Dollars
Middle East Issues: 14 hours ago, 8 flags -
Democratic Rep. Henry Cuellar and wife indicted on federal bribery charges
US Political Madness: 15 hours ago, 6 flags -
SEC shuts down Trump Media auditor, BF Borgers, over fraud
Mainstream News: 16 hours ago, 5 flags -
An AI-controlled fighter jet took the Air Force leader for a historic ride
Military Projects: 15 hours ago, 4 flags -
Zionists of ATS assemble
Political Issues: 4 hours ago, 4 flags
active topics
-
UN Estimates Rebuilding Gaza Will Cost Up To 40 Billion Dollars
Middle East Issues • 47 • : crayzeed -
For Votes - President BIDEN Opens ObamaCare Health Ins to Illegal Aliens Eff Nov 1st 2024.
2024 Elections • 16 • : Disgusted123 -
Leading Surgeon from Al-Shifa Hospital Dies in Israeli Custody
Middle East Issues • 2 • : nugget1 -
Poll - Catholic Support Swings to Trump By Significant Margin
2024 Elections • 28 • : andy06shake -
Wounded Orangutan seen using a Plant as Medicine for the First Time
Fragile Earth • 24 • : andy06shake -
The Acronym Game .. Pt.3
General Chit Chat • 7789 • : bally001 -
Zionists of ATS assemble
Political Issues • 9 • : cherokeetroy -
-@TH3WH17ERABB17- -Q- ---TIME TO SHOW THE WORLD--- -Part- --44--
Dissecting Disinformation • 790 • : daskakik -
An Implausible New Electoral Process?
Political Ideology • 32 • : chr0naut -
Post A Funny (T&C Friendly) Pic Part IV: The LOL awakens!
General Chit Chat • 7152 • : Elentari
